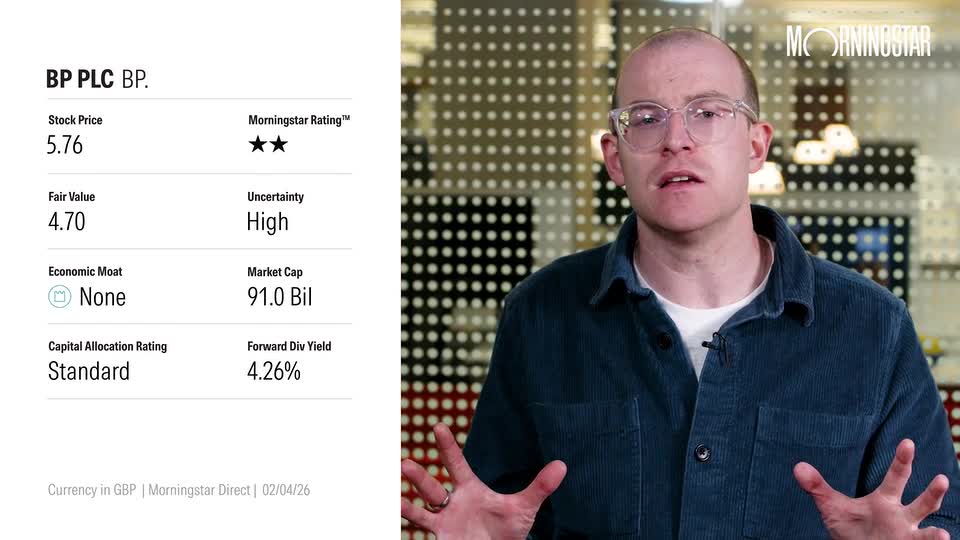
(Alliance News) - Creo Medical Group PLC on Tuesday said that it has reached its first in-human milestone for MicroBlate Flex, as part of a Lung Tissue ablation clinical study.
Creo Medical is a Chepstow, Wales-based medical device company focused on surgical endoscopy. Its MicroBlate Flex device is a new soft tissue ablation tool, which is designed to ablate nodules and tumours in several tissue types.
The first in-human use of MicroBlate Flex was successfully completed at Royal Brompton Hospital, part of Guy's and St Thomas' NHS Foundation Trust.
This procedure was part of a multi-site clinical trial, with all patients having regular follow-up assessments post-procedure to monitor their progress.
"Unlike most other cancers; there are no significant, accurate screening programmes in place for lung cancer but with screening, diagnosis and navigation technology advancing dramatically over recent years, we are set to see that change," said Chief Executive Officer Craig Gulliford.
"The whole team at Creo are delighted that this clinical program has now started. The first enrolment and MicroBlate Flex's first procedure safely completed is an important milestone in our goal to use advanced energy to transform lives."
Creo Medical shares were trading 0.04% higher at 24.26 pence each in London on Tuesday morning.
By Holly Beveridge, Alliance News reporter
Comments and questions to newsroom@alliancenews.com
Copyright 2023 Alliance News Ltd. All Rights Reserved.